The global energy storage industry has witnessed rapid expansion, yet not all battery energy storage system manufacturers deliver the same value. As demand for reliable and sustainable power grows, buyers increasingly scrutinise which solar battery storage manufacturers offer genuine differentiation. From technological depth to global reach, market leaders distinguish themselves through innovation, scalability, and long-term service commitment.
Technological Innovation and Digital Energy Evolution
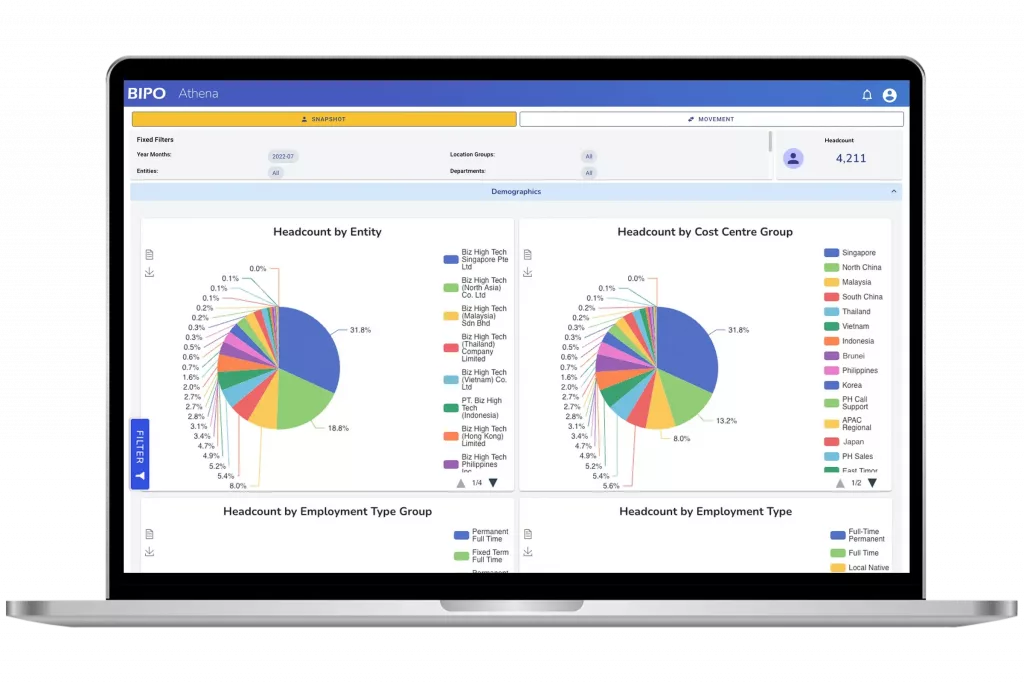
Top-tier manufacturers invest heavily in R&D to improve energy density, cycle life, and safety. Dyness exemplifies this approach. Specialising in the research, development, production, and sales of energy storage systems, Dyness continuously enhances its innovation capabilities to introduce market-ready solutions covering commercial, industrial, and residential applications. Guided by its brand mission of “Driving digital energy evolution to deliver sustainable energy for all”, Dyness stands out among solar battery storage manufacturers by integrating digital intelligence into battery management, enabling higher efficiency and smarter energy use.
Global Presence and Localised Service Advantages
A key differentiator is the ability to serve diverse markets reliably. Leading battery energy storage system manufacturer operates across continents while adapting to local regulations and grid conditions. Dyness has distributed its products across more than 100 countries and regions, delivering premium energy storage solutions to over 1,000,000 users worldwide. By actively advancing its brand strategy of “Globalization, Tech Development, and Service Advantages,” Dyness ensures that clients receive not only high-performance hardware but also responsive technical support and lifecycle management.
Summing Up the Competitive Edge
What ultimately separates market leaders from the rest is the combination of deep technical expertise, global scalability, and user-centric service. As demand for battery energy storage system manufacturers intensifies, those like Dyness, which balance innovation with real-world deployment across millions of users, set the benchmark. For businesses seeking reliable partners, evaluating solar battery storage manufacturers on these three pillars—technology, global footprint, and service—remains the clearest path to long-term energy resilience.